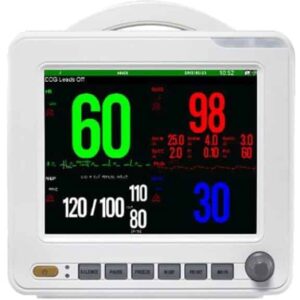
Distribute Health

VIA Rewards
Ship, Earn, Redeem

Global Logistics
Seamless Shipping Solutions

VIA Financing
Buy Now, Pay Later

Catalogue
Quality Guaranteed
VIA Rewards
Global Logistics
VIA Financing
Catalogue
Delivering Innovation to the Underserved
VIA Global Health is committed to ensuring equitable access to essential medical supplies. VIA connects underserved and remote communities with the life-saving equipment they need, fostering a world where quality healthcare is within reach for everyone, everywhere.
- Invest in a future where every mother and child has access to life-saving care.
- Expand the visibility and reach of innovative life-saving products. Ensuring that groundbreaking technologies are available to the most remote and underserved areas.
- Collaborate closely with local distributors, clinicians, and NGOs to gain insights into the unique healthcare needs of different underserved regions, ensuring that the right products are delivered to those who need them most.
Connect and Thrive: Uniting Medical Buyers and Sellers Worldwide
VIA Global Health bridges the gap in global healthcare distribution by eliminating the barriers between medical product buyers and sellers. We understand that these inefficiencies disproportionately affect underserved communities, which is why we specialize in verifying, sourcing, and shipping medical supplies to hard-to-reach areas.
Our mission is to ensure that every individual, regardless of their location, has access to quality healthcare. Whether you’re in Africa, Asia, or any other continent, we provide transparent solutions to connect you with essential medical devices and equipment on a global scale. No matter where you are or what your budget is, we are committed to bridging the gap and ensuring global access to quality healthcare.
Featured Products

The IRIS by Liger: Colposcope & Coagulator in One

Yonker Handheld Color Doppler Ultrasound – Convex and Linear

BPL Colpoview Colposcope

LifeWrap Non-Pneumatic Anti-Shock Garment (NASG)

Thermocoagulator

Keyar Echo – Smart Handheld Fetal Doppler

Warmilu IncuBlanket and InstaWarmer Kit

Pumani bubbleCPAP
How VIA Works
Connect with us by requesting a personalized quote or signing up with us and engaging in a conversation with our dedicated agent.

Discover our services and flexible payment options, including VIA Rewards and Financing, with personalized assistance from our dedicated agents.

We specialize in taking the logistical headache off your shoulders by managing every aspect ourselves.
Catalogue Preview
Organizations That Trust and Support VIA




What Our Customers Are Saying
“Over the past few years, we have been able to purchase equipment through VIA Global Health that we have found this to be very helpful, robust and reasonably priced. We are looking forward to continuing our collaboration and exploring other pieces of equipment provided through VIA. The main purpose of this message is, however, is to highlight how much we appreciate the professionalism, availability and reliability of our sales contact at VIA Global Health. He has been extraordinarily helpful and supportive.”
Why Buy from VIA?
When you choose VIA Global Health, you gain exclusive access to the latest standard of care products and cutting-edge innovations to equip your medical facility with the very best healthcare solutions for your community. With VIA, you’ll experience a multitude of advantages:
- Be the first to discover the carefully sourced products tailored to serve your specific market.
- Enjoy quick and seamless access to transparent medical equipment pricing.
- Benefit from priority support provided by our trusted sales team, who will expertly address your queries and ensure confidence at every stage of the process.
Enabling Quality Healthcare
Newborn Respiratory and Oxygen Support Products
Which newborn respiratory and oxygen support products are best for you? Visit VIA’s Buyers Guides to learn more about how these devices are used, and which one may work for you and your healthcare setting. From pulse oximeters, to innovative bCPAPs, to resuscitation devices and oxygen accessories, VIA shares expertise and offers affordable equipment and supplies in these guides.
Newborn Jaundice Products
How to choose jaundice meters and phototherapy devices is an important decision. VIA Global Health sources products to diagnose and treat newborn jaundice, and our Buyers Guides provide information and details about the condition and related products. Let us help you make a well-informed decision.
Temperature Stability Products for Newborns
Deciding between devices for treating and monitoring newborn hypothermia? VIA has incubators, radiant warmers, wraps, and temperature monitors to help with thermal protection and temperature stability for babies, and our Buyers Guides can help you choose which device will help safely care for newborns with hypothermia.
Cervical Cancer Products
Do you need products for diagnosing and treating Cervical Cancer or pre-cancerous lesions? VIA Global Health has several products to assist health care workers and women, including curettes, ferris subsulfate solution, a colposcope system, and a thermocoagulator. Learn more about Cervical Cancer and the products we offer in this Buyers Guide.